Please do not block ads on this website.
No ads = no money for us = no free stuff for you!
Electron Shell Concept
The number of electrons around the nucleus of each atom is equal to the number of protons in its nucleus.
That is, for an atom to be neutral (no overall charge), the number of negative charges (electrons) must be equal to the number of positive charges (protons).
The number of protons in the nucleus of an atom is equal to its atomic number (Z).
Therefore, the number of electrons in an atom of an element is equal to the element's atomic number (Z):
For a neutral atom:
no. electrons = no. protons = atomic number (Z)
But the atomic number (Z) only tells us how many electrons in total there are around the nucleus of a neutral atom, it doesn't directly tell us how those electrons are arranged around the nucleus.
We can model the electronic structure of atoms as a series of concentric spheres, or shells, around the nucleus of an atom.
In the diagram below, the black circle represents the nucleus of the atom, and each circle represents the boundary of an electron shell.
The shell closest to the nucleus is given the symbol K, the next shell is the L shell, which is followed by the M shell, then the N shell:
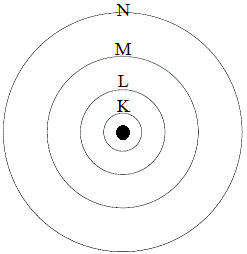
As the boundary of each shell gets further from the nucleus, the shell gets larger, so it makes sense that shells closer to the nucleus can contain fewer electrons than shells further away from the nucleus.
Indeed, we find that the K shell can contain a maximum of only 2 electrons, but the next shell, the L shell, can contain a maximum of 8 electrons.
We could draw the electronic structure of each atom by placing a dot representing an electron in each shell, but that would take up a lot of space.
So, scientists have come up with a short cut for representing the electronic structure of atoms, and this is based on an understanding of the energy of electrons in each shell.
Electrons closest to the nucleus, in the K shell, have less energy than electrons in the next shell out from the nucleus, the L shell.
Energy of electrons increases as they get further away from the nucleus. So instead of talking about "shells" we can also talk about energy levels:
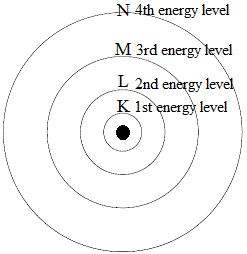
And now we can draw the energy levels only, without drawing the rest of the circle to represent the shell:
| ↑ ↑ ↑ ↑ | 3rd energy level
(M shell) | ____ | |
| ↑ ↑ | | |
| ↑ ↑ ↑ | 2nd energy level
(L shell) | ____ | (maximum of 8 electrons) |
| ↑ | | |
| ↑ | 1st energy level
(K shell) |
____ | (maximum of 2 electrons) |
↑
energy | levels
(shells) | | |
And, we can now see an ever shorter way to represent the electronic structure of an atom, we can just write down how many electrons are present in each shell (energy level) separated by a comma, and this shorthand is referred to as the electron configuration or electronic configuration of the atom.
By assuming that an atom will exist in its lowest possible energy state, we can now add the electrons one by one to the energy levels (shells).
Electrons are shown as arrows (↑ and ↓) in the diagrams below.
The first electron goes into the 1st energy level (K shell):
| ↑ ↑ ↑ ↑ | 3rd energy level
(M shell) | ____ | |
| ↑ ↑ | | |
| ↑ ↑ ↑ | 2nd energy level
(L shell) | ____ | (maximum of 8 electrons) |
| ↑ | | |
| ↑ | 1st energy level
(K shell) | ↑
____ | (maximum of 2 electrons) |
↑
energy | levels
(shells) | | |
The electron configuration (electronic configuration) of the atom is 1
If an atom has 2 electrons, then both the first and second electron occupy the 1st energy level (K shell):
| ↑ ↑ ↑ ↑ | 3rd energy level
(M shell) | ____ | |
| ↑ ↑ | | |
| ↑ ↑ ↑ | 2nd energy level
(L shell) | ____ | (maximum of 8 electrons) |
| ↑ | | |
| ↑ | 1st energy level
(K shell) | ↑↓
____ | (maximum of 2 electrons) |
↑
energy | levels
(shells) | | |
The electron (electronic) configuration of the atom is 2
K shell is full because the K shell can only hold 2 electrons, so the next electron must be positioned in the next energy level (next shell).
So an atom with 3 electrons will have 2 electrons in the K shell and the third electron goes into the 2nd energy level (L shell):
| ↑ ↑ ↑ ↑ | 3rd energy level
(M shell) | ____ | |
| ↑ ↑ | | |
| ↑ ↑ ↑ | 2nd energy level
(L shell) | ↑
____ | (maximum of 8 electrons) |
| ↑ | | |
| ↑ | 1st energy level
(K shell) | ↑↓
____ | (maximum of 2 electrons) |
↑
energy | levels
(shells) | | |
The electron configuration (electronic configuration) of the atom is 2,1
(2 electrons in the first energy level and 1 electron in the second energy level)
For atoms with more than 3 electrons, we can continue adding electrons to the second energy level or L shell until it reaches its maximum capacity of 8 electrons:
Fourth electron goes into the 2nd energy level (L shell) ...
.... until the 2nd energy level (L shell) is filled with 8 electrons.
Once both the 1st energy level (K shell) and the second energy level (L shell) are full, we must begin filling the next energy level, the 3rd energy level (M shell).
We can infer how the electrons are arranged around a nucleus by considering the position of an element in the periodic table.
The elements of the first three periods of the periodic table are shown below:
| |
Group
1 |
Group
2 |
Groups
3 - 12 |
Group
13 |
Group
14 |
Group
15 |
Group
16 |
Group
17 |
Group
18 |
Period 1:
filling K shell
(1st energy level) |
Z=1
H
hydrogen
|
|
Z=2
He
helium
|
Period 2
filling L shell
(2nd energy level) |
Z=3
Li
lithium
|
Z=4
Be
beryllium
|
|
Z=5
B
boron
|
Z=6
C
carbon
|
Z=7
N
nitrogen
|
Z=8
O
oxygen
|
Z=9
F
fluorine
|
Z=10
Ne
neon
|
Period 3
filling M shell
(3rd energy level) |
Z=11
Na
sodium
|
Z=12
Mg
magnesium
|
|
Z=13
Al
aluminium
|
Z=14
Si
silicon
|
Z=15
P
phosphorus
|
Z=16
S
sulfur
|
Z=17
Cl
chlorine
|
Z=18
Ar
argon
|
Electronic Configuration of Atoms of Period 1 Elements
There are only two elements in period 1 of the periodic table; hydrogen and helium:
| |
Group
1 |
Group
2 |
Groups
3 - 12 |
Group
13 |
Group
14 |
Group
15 |
Group
16 |
Group
17 |
Group
18 |
Period 1
filling K shell
(1st energy level) |
Z=1
H
hydrogen
|
|
Z=2
He
helium
|
An atom of hydrogen only has 1 electron, so this electron occupies the K shell (first energy level), and its shell electron configuration is simply 1
An atom of helium has 2 electrons, both of which can occupy the K shell (first energy level), so its shell electron configuration is simply 2
Name of Atom
(symbol) |
hydogen
(H) |
helium
(He) |
|---|
| Atomic Number (Z) |
1 |
2 |
|---|
| No. electrons (=Z) |
1 |
2 |
|---|
No. electrons in K shell
(first energy level) |
1 |
2 |
|---|
| shell electron configuration |
1 |
2 |
|---|
The electrons in the atoms of elements in Period 1 are filling the first energy level, or K shell.
Electronic Configuration of Atoms of Period 2 Elements
There are 8 elements in Period 2 of the periodic table:
| |
Group
1 |
Group
2 |
Groups
3 - 12 |
Group
13 |
Group
14 |
Group
15 |
Group
16 |
Group
17 |
Group
18 |
Period 2:
filling L shell
(2nd energy level) |
Z=3
Li
lithium
|
Z=4
Be
beryllium
|
|
Z=5
B
boron
|
Z=6
C
carbon
|
Z=7
N
nitrogen
|
Z=8
O
oxygen
|
Z=9
F
fluorine
|
Z=10
Ne
neon
|
The atoms of elements in Period 1 filled, or completed, the first energy level, or K shell.
The atoms of the elements of Period 2 elements all have the first energy level, K shell, full, that is, the electron configuration begins with a 2
The atoms of the elements in Period 2 must be filling the second energy level, or L shell.
The number of electrons in the second energy level (L shell) is separated from those in the first by a comma when writing the electron configuration (electronic configuration).
The beginning of the electron configuration (electronic configuration) of all period 2 elements will be 2,
Name of Atom
(symbol) |
lithium
(Li) |
beryllium
(Be) |
boron
(B) |
carbon
(C) |
nitrogen
(N) |
oxygen
(O) |
fluorine
(F) |
neon
(Ne) |
|---|
| Atomic Number (Z) |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
|---|
| No. electrons (=Z) |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
|---|
No. electrons in K shell
(first energy level) |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
|---|
No. electrons in L shell
(second energy level) |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
|---|
| shell electron configuration |
2,1 |
2,2 |
2,3 |
2,4 |
2,5 |
2,6 |
2,7 |
2,8 |
|---|
The electron configuration (electronic configuration) of carbon is 2,4
This tells us that an atom of carbon has 2 electrons in the first energy level (K shell), and 4 electrons in the second energy level (L shell).
The total number of electrons in an atom of carbon is equal to the sum of all the electrons in all the energy levels (shells):
total number of electrons in a carbon atom = 2 + 4 = 6
and the total number of electrons in an atom of carbon is equal to its atomic number (Z), 6, which tells us the number of protons in the nucleus of an atom of carbon.
The maximum number of electrons that can be placed in the second energy level (L shell) is 8.
This means that atoms of neon have a full K shell (first energy level) and a full L shell (second energy level).
Electronic Configuration of Atoms of Period 3 Elements
There are 8 elements making up period 3 of the periodic table:
| |
Group
1 |
Group
2 |
Groups
3 - 12 |
Group
13 |
Group
14 |
Group
15 |
Group
16 |
Group
17 |
Group
18 |
Period 3:
filling M shell
(3rd energy level) |
Z=11
Na
sodium
|
Z=12
Mg
magnesium
|
|
Z=13
Al
aluminium
|
Z=14
Si
silicon
|
Z=15
P
phosphorus
|
Z=16
S
sulfur
|
Z=17
Cl
chlorine
|
Z=18
Ar
argon
|
Electrons of atoms of period 1 elements were filling the first energy level (K shell).
Electrons of atoms of period 2 elements had a completed first energy level (K shell) and were filling up the second energy level (L shell), which was completed by the last member of the period, neon.
Atoms of period 3 elements must:
- have full K shells (2 electrons in the first energy level)
- have full L shells (8 electrons in the second energy level)
- be filling the M shell (third energy level)
So the electron configuration (electronic configuration) of all period 3 elements must begin with 2,8
Name of Atom
(symbol) |
sodium
(Na) |
magnesium
(Mg) |
aluminium
(Al) |
silicon
(Si) |
phosphorus
(P) |
sulfur
(S) |
chlorine
(Cl) |
argon
(Ar) |
|---|
| Atomic Number (Z) |
11 |
12 |
13 |
14 |
15 |
16 |
17 |
18 |
|---|
| No. electrons (=Z) |
11 |
12 |
13 |
14 |
15 |
16 |
17 |
18 |
|---|
No. electrons in K shell
(first energy level) |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
|---|
No. electrons in L shell
(first energy level) |
8 |
8 |
8 |
8 |
8 |
8 |
8 |
8 |
|---|
No. electrons in M shell
(second energy level) |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
|---|
| shell electron configuration |
2,8,1 |
2,8,2 |
2,8,3 |
2,8,4 |
2,8,5 |
2,8,6 |
2,8,7 |
2,8,8 |
|---|
The electron configuration (electronic configuration) of an atom of sulfur is 2,8,6
An atom of sulfur has 2 electrons in the first energy level (K shell), 8 electrons in the second energy level (L shell) and 6 electrons in the third energy level (M shell).
The total number of electrons in an atom of sulfur = 2 + 8 + 6 = 16
which is the same as sulfur's atomic number (Z = 16), which means the number of protons in an atom of sulfur (16) equals the number of electrons in an atom of sulfur (16).


