| The infrared (ir) spectrum of hexane, CH3CH2CH2CH2CH2CH3. |
Bond types present - C-H stretches
(2850-3300 cm-1)
|
 |
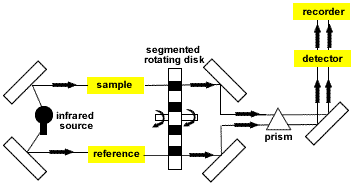
| The infrared (ir) spectrum of hex-1-ene (1-hexene), H2C=CHCH2CH2CH2CH3. |
Bond types present - C-H stretches
(2850-3300 cm-1) - C=C stretches
(1610-1680 cm-1)
|
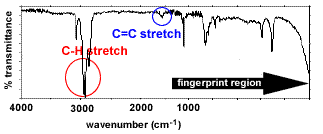 |
| The infrared (ir) spectrum of ethanol, CH3CH2OH. |
Bond types present - O-H (alcohol) stretches
(3200-3550 cm-1) - C-H stretches
(2850-3300 cm-1)
|
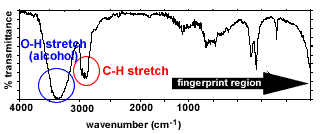 |
| The infrared (ir) spectrum of octanal, | CH3CH2CH2CH2CH2CH2CH2 | O
||
C | H |
|
Bond types present - C-H stretches
(2850-3300 cm-1) - C=O stretches
(1670-1750 cm-1)
|
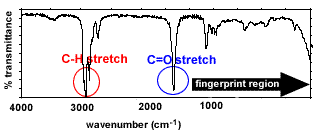 |
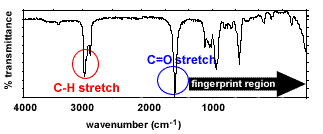
|
Bond types present - C-H stretches
(2850-3300 cm-1) - C=O stretches
(1670-1750 cm-1)
|
 |