Footnotes:
(1) For this tutorial, we will only be concerned with examples where R is an alkyl group (that is a group derived from the alkane series).
And, we only be dealing with esters derived from straight chain carboxylic acids, so the name of the ester will be two words.
It should be noted that the names of more complex esters will be made up of more than 2 words.
If more than organyl group (alkyl, aryl etc) is present, they are cited in alphabetical order.
(2) During the esterification reaction, the C-O bond in the carboxylic acid is broken.
Evidence for this comes from experiments using isotopically labeled reactants.
If some of the alcohol used contains the 18O isotope, the atoms of this isotope are all found in the ester product and NOT in the water.
(3) There are other ways to produce esters besides the direct (Fischer) esterfication of a carboxylic acid with an alcohol.
(a) Alcoholysis of acid chlorides, anhydrides, or nitriles, produces esters.
(b) Reaction of the salt of a carboxylic acid with an alkyl halide or sulfate will produce an ester.
(c) Trans-esterification of an ester produces a new ester.
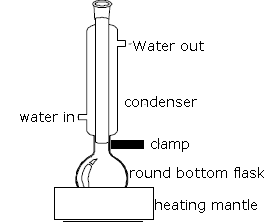
(4) If a liquid is largely free of air, and the glass flask is clean and very smooth, the liquid may superheat, a condition in which the temperature of the liquid rises above its boiling point.
In a superheated liquid it is hard to form vapour bubbles resulting in irregular expulsion of bubbles of vapour, or, bumping.
Boiling chips (pieces of porous pot) provide an additional source of minute air bubbles which act as a nucleus for building bubbles of vapour in the liquid, allowing the liquid to boil quietly.
(5) An even better way to improve ester yield is to use anhydrides in the synthesis instead of carboxylic acids.
For example, the reaction between ethanol and acetic anhydride is irreversible, and goes to completion within minutes.
acetic anhydride + ethanol → ethyl acetate + acetic acid
(6) Although sulfuric acid plays a vital role in the esterification reaction mechanism, it is beyond the scope of this tutorial. Suffice it to say that since the sulfuric acid that is used during in the reaction is re-produced at the end of the reaction mechanism, sulfuric acid is acting as a catalyst for the reaction.
(7) For a saturated sodium carbonate solution, dissolve 4.5 g sodium carbonate in 15 mL of distilled water.
(8) If you are an organic chemist you will name HO-CO-O-Na+ as sodium hydrogen carbonate, but if you are an inorganic chemist you will probably name it sodium hydrogencarbonate.
Another common name for the same compound is sodium bicarbonate.
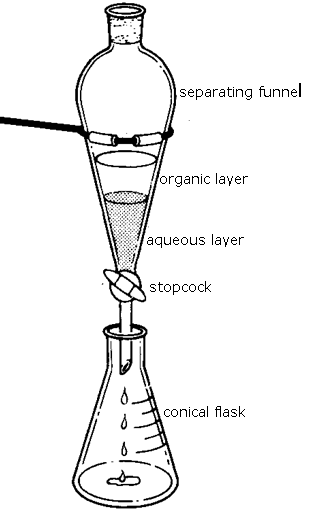
(9) Anhydrous magnesium sulfate is used as a drying agent in preference to fused or "anhydrous" calcium chloride which can combine with some esters.
Note the use of the term "drying agent". No chemical change is involved in using a drying agent, the drying agent is used to remove excess water molecules from the mixture.
In a dehydration reaction, however, there is a chemical change. Atoms of hydrogen and oxygen are removed from reactant molecules and produce water as product.