Please do not block ads on this website.
No ads = no money for us = no free stuff for you!
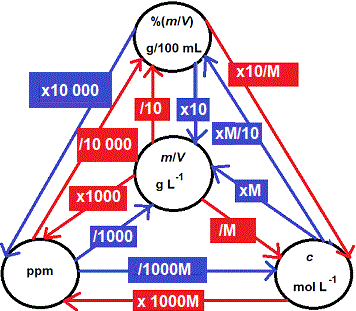
How to Convert Between Concentration Units
We can report the concentration of a solution in a number of different ways based on the volume of the solution:
⚛ mass concentration
symbol: m/V or w/v
units: g mL-1 or g L-1
⚛ mass-volume percentage concentration
symbol: %(m/V) or %(w/V)
units: g/100 mL or %
⚛ parts per million
symbol: ppm
units: mg L-1 or μg mL-1
⚛ amount of substance concentration (molarity)
symbol: c
units: mol L-1 (or M)
Note how the amount of substance concentration (molarity) is calculated:
| c = |
moles of solute
volume of solution |
= |
n
V |
| c = |
mass of solute/molar mass of solute
volume of solution |
= |
m/M
V |
Can you see that ALL of the concentration units above are based on:
Therefore we can use the mass concentration (m/V or w/V) as the basis for converting between all these different units.(2)
If, for example, we make an aqueous solution of copper(II) sulfate, CuSO(aq), by dissolving 0.250 g of solid copper(II) sulfate, CuSO4(s), in enough water to make 250.0 mL of solution, then the mass concentration of the solution can be calculated:
mass concentration = m/V = 0.250 g ÷ 250.0 mL = 0.00100 g mL-1
and because there are 1000 mL in 1 L, we could also determine mass concentration in units of grams of solute per litre of solution:
mass concentration = 0.00100 g mL-1 × 1000 mL/L = 1.00 g L-1
In general:
1000 × (m/V g mL-1) = m/V g L-1
Mass-volume percentage concentration, %(m/V), has the units grams of solute per 100 mL of solution.
So we can convert mass concentration in g mL-1 to g/100 mL by multiplying the mass and the volume in the mass concentration by 100:
| %(m/V) = |
m (g) × 100
V (mL) × 100 |
For our copper(II) sulfate solution with a mass concentration of 0.00100 g mL-1, we can calculate the concentration as a mass-volume percentage concentration as shown below:
| %(m/V) = |
m (g) × 100
V (mL) × 100 |
= |
0.00100 (g) × 100
1 (mL) × 100 |
= |
0.100 g
100 mL
|
Note that the mass-volume percentage concentration may be given as 0.100 g/100 mL or as 0.100 %(m/V)
In general:
100 × (m/V g mL-1) = %(m/V) or g/100 mL
The units for parts per million concentration (ppm) are μg mL-1.
There are 1 million micrograms in a gram, 1 000 000 μg = 1 g
So we can convert mass concentration in g mL-1 to μg mL-1 by multiplying the mass ONLY by 1 000 000 :
| ppm = |
m (g) × 1 000 000 μg/g
V (mL) |
For our copper(II) sulfate solution with a mass concentration of 0.00100 g mL-1, we can calculate the concentration as a μg L-1 as shown below:
| ppm = |
m (g) × 1 000 000 μg/g
V (mL) |
= |
0.00100 g × 1 000 000 μg/g
1 mL |
= |
1000 μg
1 mL |
The concentration could be recorded as 1000 μg mL-1 or as 1000 ppm.
Because there are 1000 mL in 1 L and 1000 μg in 1 mg, we could convert this parts per million concentration in μg of solute per mL of solution to a parts per million concentration in mg of solute per litre of solution by dividing the mass in μg by 1000 and dividing the volume in mL by 1000.
So we can convert the concentration of our copper(II) sulfate solution from μg mL-1 to mg L-1 as shown below:
| ppm = |
= |
1000 μg ÷ 1000 μg/mg
1 mL ÷ 1000 mL/L |
= |
1 mg
0.001 L |
= |
1000 mg
1 L |
The concentration of our copper(II) sulfate solution could therefore be reported as 1000 mg L-1 or 1000 ppm
In general:
1 000 000 × (m/V g mL-1) = ppm (either μg mL-1 or mg L-1)
The units for amount of substance concentration, molarity, are moles of solute per litre of solution.
Moles of solute is calculated by dividing the mass of the solute in grams by the molar mass of the solute in grams per mol:
n(solute) mol = m(solute) g ÷ M(solute) g mol-1
We can use the periodic table to find the atomic weight (atomic mass) of each element and use this to calculate the molar mass of the solute (relative molecular mass expressed in grams).
For CuSO4:
M(CuSO4) = Mr(Cu) + Mr(S) + 4×Mr(O)
M(CuSO4) = 63.55 + 32.06 + 4×16.00 = 159.61 g mol-1
Mass concentration can be expressed in units of g L-1.
Our copper(II) sulfate solution has a mass concentration of 1 g L-1.
We can convert this to mol L-1 by dividing the mass of solute by its molar mass :
| c mol L-1 = |
= |
m g/Mr g mol-1
V L |
= |
1 g/159.61 g mol-1
1 L |
= |
0.00627 mol L-1 |
In general:
1 000 × [(m ÷ Mr)/V] g mL-1 = c mol L-1
[(m ÷ Mr)/V] g L-1 = c mol L-1
It is worth noting the special relationship between %(m/V) and ppm :
%(m/V) gives us the concentration in parts of solute per 100 parts of solution (that is, a percentage)
ppm gives us the concentration in parts of solute per 1 000 000 parts of solution (that is, parts per million)
Therefore, to convert %(m/V) to ppm, we multiply it by a factor of 1 000 000/100 = 10 000
The %(m/V) of our CuSO4(aq) is 0.1 g/100 mL (or 0.001 %) so its concentration in ppm is:
ppm = 10 000 × %(m/V) = 10 000 × 0.1 g/100 mL = 1000 ppm
In general:
10 000 × %(m/V) = ppm (either mg L-1 or μg mL-1)
It is also worth noting that a mass concentration (m/V) given in units of g L-1 could also be expressed as g/1000 mL, or parts per thousand.
So to convert a parts per thousand concentration, g L-1, to parts per hundred, %(m/V), we only need to divide by 10: (m/V) ÷ 10 = %(m/V)
To convert a parts per thousand concentration, g L-1 or g/1000 mL, to parts per million, we just multiply by 1000: (m/V) × 1000 = ppm (either μg mL-1 or mg L-1)
You should practise converting one concentration unit to another, for example, how would you convert parts per million (ppm) to an amount of substance concentration (molarity) ?
You can check your calculations using the summary table in the next section.
↪ Back to top
Worked Examples: Converting Concentration Units
Question 1: The mass concentration of citric acid in a sample of orange juice was 45 g L-1.
What is the concentration of citric acid in this orange juice in parts per million ?
Solution:
Step 1: Extract the data from the question:
m/V(citric acid) = 45 g L-1
ppm = ?
Step 2: What is the relationship between what you know and what you need to find out?
1 ppm = 1 mg L-1
1000 mg = 1 g
m/V(g L-1) × 1000 mg/g = 1000 × m/V = ppm (mg L-1)
Step 3: Perform the calculations:
m/V(45 g L-1) × 1000 mg/g = 45 000 mg L-1
1 ppm = 1 mg L-1
45 000 mg L-1 = 45 000 ppm
Question 2: The concentration of oxygen gas dissolved in a water sample is 6.5 ppm.
What is the mass-volume percentage concentration of oxygen gas in this water sample ?
Solution:
Step 1: Extract the data from the question:
concentration(oxygen gas) = 6.5 ppm
%(m/V) = ? g/100 mL
Step 2: What is the relationship between what you know and what you need to find out?
1 ppm = 1 μg mL-1
1 μg mL-1 ÷ 1 000 000 μg/g = 1 g mL-1
(100 × 1) g ÷ (100 × 1 mL) = %(mV) g/100 mL
Step 3: Perform the calculations:
6.5 ppm = 6.5 μg mL-1
6.5 μg mL-1 ÷ 1 000 000 μg/g = 6.5×10-6 g mL-1
%(mV) = (100 × 6.5×10-6) g ÷ (100 × 1 mL) = 6.5×10-4 g/100 mL = 0.00065 %(m/V)
Question 3: Atheltes often consume a "sports drink", an aqueous solution of electrolytes like chloride ions, to replace ions that they lost in sweat.
One sample of "sports drink" had a chloride ion concentration of 675 ppm.
Calculate the amount concentration (molarity) of chloride ions in this "sports drink".
Solution:
Step 1: Extract the data from the question:
concentration(Cl-(aq)) = 675 ppm
c(Cl-(aq)) = ? mol L-1
Step 2: What is the relationship between what you know and what you need to find out?
1 ppm = 1 mg L-1
1 g = 1000 mg
n(Cl-(aq)) = (m(Cl-(aq) in mg/1000 mg/g) ÷ M(Cl-(aq)) = ? mol
c(Cl-(aq)) = n(Cl-(aq)) ÷ V
Step 3: Perform the calculations:
675 ppm = 675 mg L-1
n(Cl-(aq)) = (675 mg/1000 mg/g) ÷ 35.45 g mol-1 = 0.0190 mol
c(Cl-(aq)) = 0.0190 mol ÷ 1 L = 0.0190 mol L-1
↪ Back to top